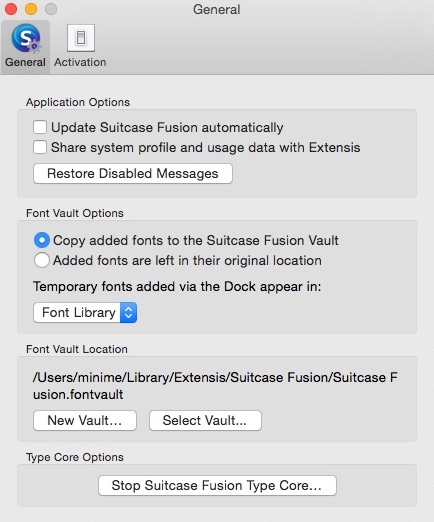
The fusion loops on one monomer are indicated by a red asterisk (33). The lipid bilayers are drawn as gray bars the viral membranes are along the bottom of the figure, and the cell membranes, along the top. C-terminal ectodomain residues missing from the crystal structures are drawn as thick lines transmembrane residues, as rods. The G monomers are colored blue, green, and yellow, respectively. G is a trimer in both its prefusion and postfusion states. Proposed pathway of sequential conformational changes in G that drive membrane fusion. Our study of VSV now extends this description to "class III" viral fusion proteins, showing that reversibility of the low-pH-induced transition and architectural differences in the fusion proteins themselves do not change the basic mechanism by which they catalyze membrane fusion.Įnveloped virus membrane fusion protein virus entry. This sequence of conformational events is similar to those shown to describe fusion by influenza virus hemagglutinin (a "class I" fusogen) and West Nile virus envelope protein ("class II"). From simulations of the kinetic data, we conclude that the critical number of G-protein trimers required to overcome membrane resistance is 3 to 5, within a contact zone between the virus and the target membrane of 30 to 50 trimers. We show that a three-stage model fits VSV single-particle fusion kinetics: (i) reversible, pH-dependent, G-protein conformational change from the known prefusion conformation to an extended, monomeric intermediate (ii) reversible trimerization and clustering of the G-protein fusion loops, leading to an extended intermediate that inserts the fusion loops into the target-cell membrane and (iii) folding back of a cluster of extended trimers into their postfusion conformations, bringing together the viral and cellular membranes. We report single-virion fusion experiments, using methods developed in previous publications to probe fusion of influenza and West Nile viruses. The reversibility of their fusogenic conformational transitions differentiates them from many other low-pH-induced viral fusion proteins.
The glycoproteins (G proteins) of vesicular stomatitis virus (VSV) and related rhabdoviruses (e.g., rabies virus) mediate both cell attachment and membrane fusion.
